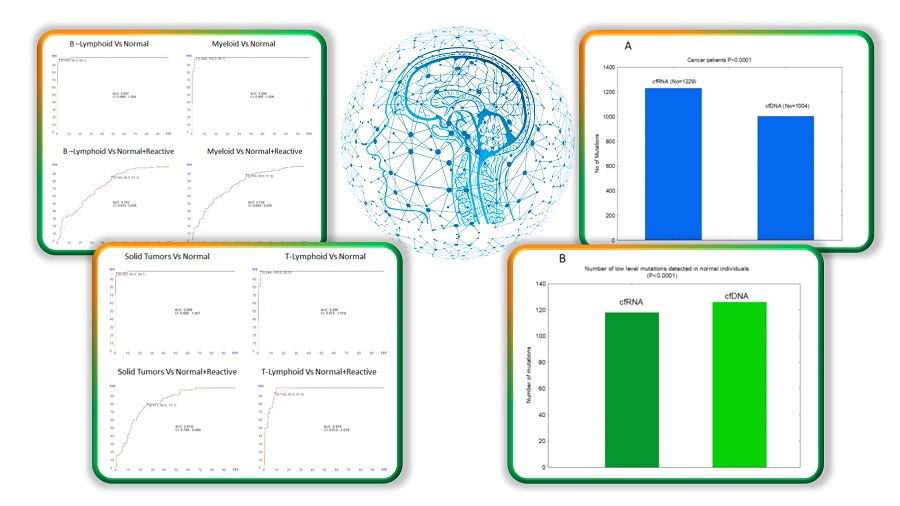
New publication on how cfRNA and AI are advancing LBx
GTC has a new publication in Heliyon titled “The Potential of Cell-Free RNA in Liquid Biopsy: Comprehensive Analysis of Mutation Profile, Chromosomal Abnormalities, and Immune

GTC has a new publication in Heliyon titled “The Potential of Cell-Free RNA in Liquid Biopsy: Comprehensive Analysis of Mutation Profile, Chromosomal Abnormalities, and Immune

Baltimore, MD and Irvine, CA, April 13, 2023, Sysmex Inostics Inc., a subsidiary of Japan’s Sysmex Corporation and Baltimore-based biotechnology firm, and Genomic Testing Cooperative

Demonstrating Reliability of Targeted Transcriptomic Profiling in Predicting Levels of HER2, ER, PD-L1 and Other Immunohistochemistry-based Biomarkers when Combined with Artificial Intelligence Irvine, California– April
CHICAGO – With the publication of a paper validating two artificial intelligence-based algorithms for assisting pathologists in molecular profiling of patients, the Genomic Testing Cooperative

GTC research is being presented at ASCO 2020 in the following three abstracts. 1. FGFR expression, fusion and mutation as detected by NGS sequencing of

GTC featured in the Orange County Business Journal on September 30, 2019 [Click image to enlarge]
MDS is defined as ineffective hematopoiesis characterized by adequate or increased hematologic elements in bone marrow with peripheral cytopenia. Therefore, the presence of cytopenia in
Genomic Testing cooperative (GTC) provides a comprehensive genomic profiling of breast cancer including evaluating: – Actional mutations – Gene amplification and deletion – Hormonal expression

(FOR UPDATED INFORMATION ABOUT COMPREHENSIVE TESTING FOR BREAST CANCER VISIT THIS PAGE: Comprehensive testing for breast cancer evaluating DNA and RNA) When you order GTC
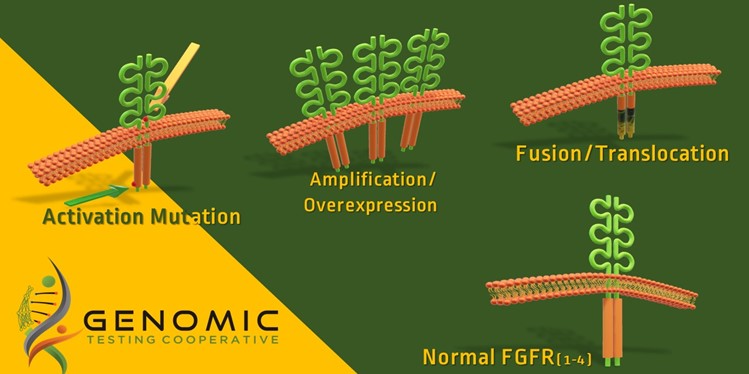
The FDA has approved Balversa (erdafitinib) as a treatment for adult patients with locally advanced or metastatic bladder cancer that have FGFR3 or FGFR2 abnormalities