GTC scientists collaborate with the NIH on a study just published in the Journal of Clinical Oncology. The publication is titled: Prediction of outcome in CLL patients treated with ibrutinib: development and validation of a four-factor prognostic model. You can read the article here (subscription required).
Abstract:
PURPOSE
Randomized trials established the superiority of ibrutinib-based therapy over chemoimmunotherapy in chronic lymphocytic leukemia. Durability of progression-free survival (PFS) with ibrutinib can vary by patient subgroup. Clinical tools for prognostication and risk-stratification are needed.
PATIENTS AND METHODS
Patients treated with ibrutinib in phase II and III trials provided the discovery data set and were subdivided into discovery and internal validation cohorts. An external validation cohort included 84 patients enrolled in our investigator-initiated phase II trial. Univariable analysis of 18 pretreatment parameters was performed using PFS and overall survival (OS) end-points. Multivariable analysis and machine-learning algorithms identified four factors for a prognostic model that was validated in internal and external cohorts.
RESULTS
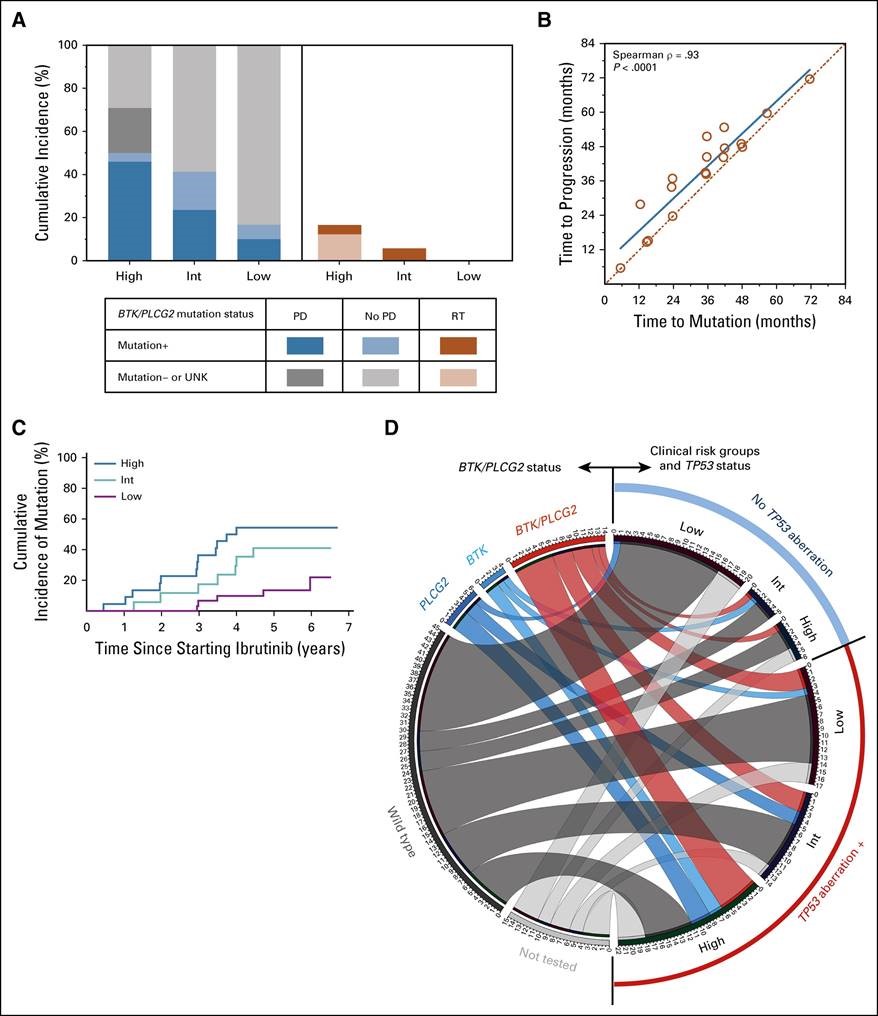
Factors independently associated with inferior PFS and OS were as follows: TP53 aberration, prior treatment, β-2 microglobulin ≥ 5 mg/L, and lactate dehydrogenase > 250 U/L. Each of these four factors contributed one point to a prognostic model that stratified patients into three risk groups: three to four points, high risk; two points, intermediate risk; zero to one point, low risk. The 3-year PFS rates for all 804 patients combined were 47%, 74%, and 87% for the high-, the intermediate-, and the low-risk group, respectively (P < .0001). The 3-year OS rates were 63%, 83%, and 93%, respectively (P < .0001). The model remained significant when applied to treatment-naïve and relapsed/refractory cohorts individually. For 84 patients in the external cohort, BTK and PLCG2 mutations were tested cross-sectionally and at progression. The cumulative incidences of mutations were strongly correlated with the model. In the external cohort, Richter’s transformation occurred in 17% of the high-risk group, and in no patient in the low-risk group.
CONCLUSION
Patients at increased risk of ibrutinib failure can be identified at treatment initiation and considered for clinical trials.
Inhye E. Ahn, MD1; Xin Tian, PhD2; David Ipe, MS3; Mei Cheng, PhD3; Maher Albitar, MD4,5; L. Claire Tsao, PhD3; Lei Zhang, MA3; Wanlong Ma, MS4,5; Sarah E. M. Herman, PhD1; Erika M. Gaglione, BA1; Susan Soto, MSN1; James P. Dean, MD, PhD3; and Adrian Wiestner, MD, PhD1
1Hematology Branch, National Heart, Lung, and Blood Institute, National Institutes of Health, Bethesda, MD
2Office of Biostatistics Research, National Heart, Lung, and Blood Institute, National Institutes of Health, Bethesda, MD
3Pharmacyclics LLC, an AbbVie Company, Sunnyvale, CA
4NeoGenomics Laboratories, Irvine, CA
5Genomic Testing Cooperative, Irvine, CA
© 2020 by American Society of Clinical Oncology


